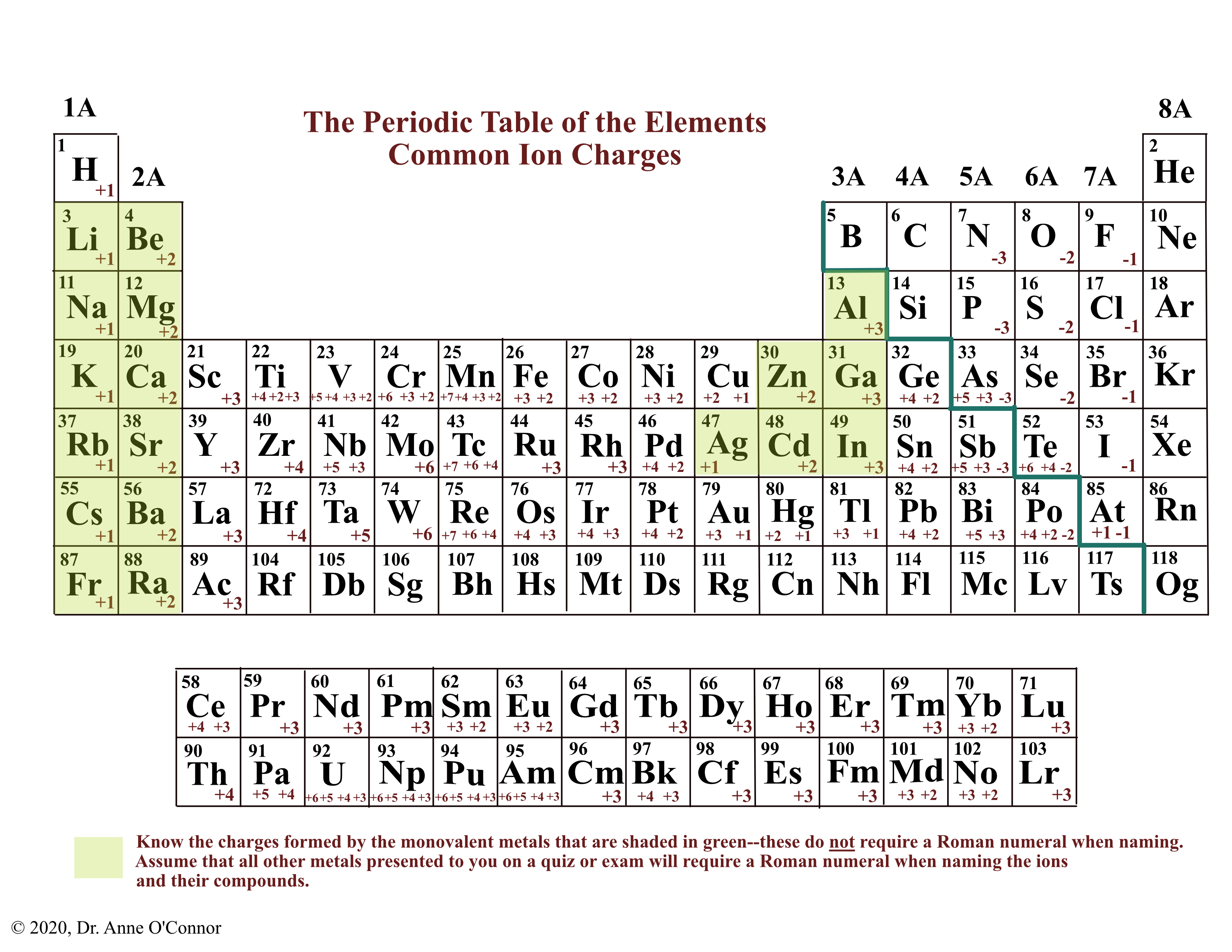
For example, potassium oxide is made up of potassium ions K + atom.The numbers of ions in a formula must give an equal number of positive and negative charges. (If it had a different charge, it would pull in another atom.) Find the number of atoms of each element that combine to "cancel out" each other's charge and make a neutral compound. Ionic Compound Formula Sodium chloride: NaCl, with Na+ and Cl ions Lithium nitride: Li3N, with Li+ and N3- ions Magnesium oxide: MgO, with Mg2+ and O2- ions. The formula of an ionic compound can be predicted by using the formulae of its ions. In this video, we'll walk through this process for the ionic compound calcium bromide. As you can see, there are no individual NaCl particles in the array instead, there is a continuous lattice of alternating sodium and chloride ions. Finally, combine the two ions to form an electrically neutral compound. Ionic compounds can also contain molecules which bear charges, and these units are charged either positively or negatively. Ionic compounds exist as alternating positive and negative ions in regular, three-dimensional arrays called crystals (Figure 2.4.1 ). Then, identify the anion and write down its symbol and charge. Ionic formulae Ionic compounds contain positive and negative ions. Can be recognized from formula if it has a combination of : Metal and a nonmetal ion, e.g. It lacks a boiling point because of this. It rapidly decomposes into its constituent elements according to the equation: 2Na3N 6Na +N2. Taken as a whole, the ionic compound is electrically neutral, meaning it has a total charge of zero. To find the formula of an ionic compound, first identify the cation and write down its symbol and charge. Therefore, the formula of sodium nitride is Na3N. Electrical forces hold together ionic compounds, pulling the positive and negative ions together. It’s crucial to balance the charges in order to write ionic compounds correctly.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
